Many assume that placing pharmaceuticals in a standard refrigerator is enough to keep them safe during transit. It is not. Temperature excursions can degrade sensitive biologics within minutes, rendering entire shipments clinically useless and potentially harmful to patients. For healthcare logistics managers operating across Southeast Asia, where geography, climate, and infrastructure vary sharply, the stakes are even higher. This guide examines what a robust cold chain actually requires, where failures occur, and how rigorous compliance protects both patients and business viability.
Table of Contents
- What is a cold chain and why does it matter?
- Key temperature ranges and pharmaceutical requirements
- Risks, edge cases, and compliance: Where the cold chain breaks
- Global standards and Southeast Asia’s regulatory landscape
- Why infrastructure, not just protocol, determines outcomes
- How to secure your cold chain: Strategic steps forward
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Unbroken chain is vital | Any lapse in temperature control can compromise medicine safety and patient outcomes. |
| Strict standards required | Global and regional regulations demand precise monitoring, mapping, and qualified equipment at every stage. |
| Edge cases are common | Last-mile delivery, rural infrastructure, and power outages are frequent sources of failure in Southeast Asia. |
| Infrastructure investment pays | Real resilience comes from modern technology and reliable infrastructure, not just compliance paperwork. |
What is a cold chain and why does it matter?
A cold chain is a continuous, temperature-controlled supply chain that preserves the integrity of pharmaceutical products from the point of manufacture all the way to patient administration. Every link matters. A warehouse, a transport vehicle, a customs holding area, a pharmacy refrigerator — each one must meet defined temperature requirements without interruption.
Cold chain logistics in Southeast Asia covers a broad category of products, including:
- Vaccines (live-attenuated and inactivated)
- Biologics such as insulin and growth hormones
- Monoclonal antibodies (mAbs) used in oncology and autoimmune therapies
- Plasma-derived medicines
- Gene therapy products
Product integrity for these temperature-sensitive pharmaceuticals depends entirely on an unbroken cold chain. The moment a product is exposed to an out-of-range temperature, molecular degradation can begin. Proteins unfold. Emulsions separate. Potency drops. These changes are often irreversible and invisible to the naked eye, meaning a compromised product may look perfectly fine but deliver no therapeutic benefit — or worse, cause harm.
A common misconception is that a standard household or commercial refrigerator provides adequate protection. It does not. Industrial cold chain infrastructure requires validated equipment, continuous data logging, alarm systems, and qualified personnel who can respond immediately to deviations.
Where the cold chain most commonly fails:
| Failure point | Common cause | Impact |
|---|---|---|
| Distribution centers | Equipment breakdown, power loss | Product spoilage across batches |
| In-transit | Inadequate insulation, delays | Excursion during longest exposure window |
| Last-mile delivery | Uncontrolled vehicles, rural access | High-risk for fragile biologics |
| Storage at point of care | Staff training gaps, overloaded fridges | Localized but high-consequence failures |
Pro Tip: Map every temperature-sensitive touchpoint in your supply chain before auditing equipment. You cannot protect what you have not identified.
Having established why the cold chain cannot be taken for granted, it is essential to examine the precise temperature requirements that govern pharmaceutical storage.
Key temperature ranges and pharmaceutical requirements
Pharmaceuticals are not one-size-fits-all when it comes to storage. Regulatory standards define distinct temperature bands, and even a brief deviation outside these ranges can trigger irreversible molecular changes.

Standard pharmaceutical storage temperature ranges:
| Temperature range | Category | Typical products |
|---|---|---|
| 15°C to 25°C | Controlled Room Temperature (CRT) | Tablets, capsules, topical creams |
| 2°C to 8°C | Refrigerated | Vaccines, insulin, most mAbs |
| -20°C | Frozen | Some vaccines, plasma products |
| Below -80°C | Cryogenic | mRNA vaccines, advanced biologics |
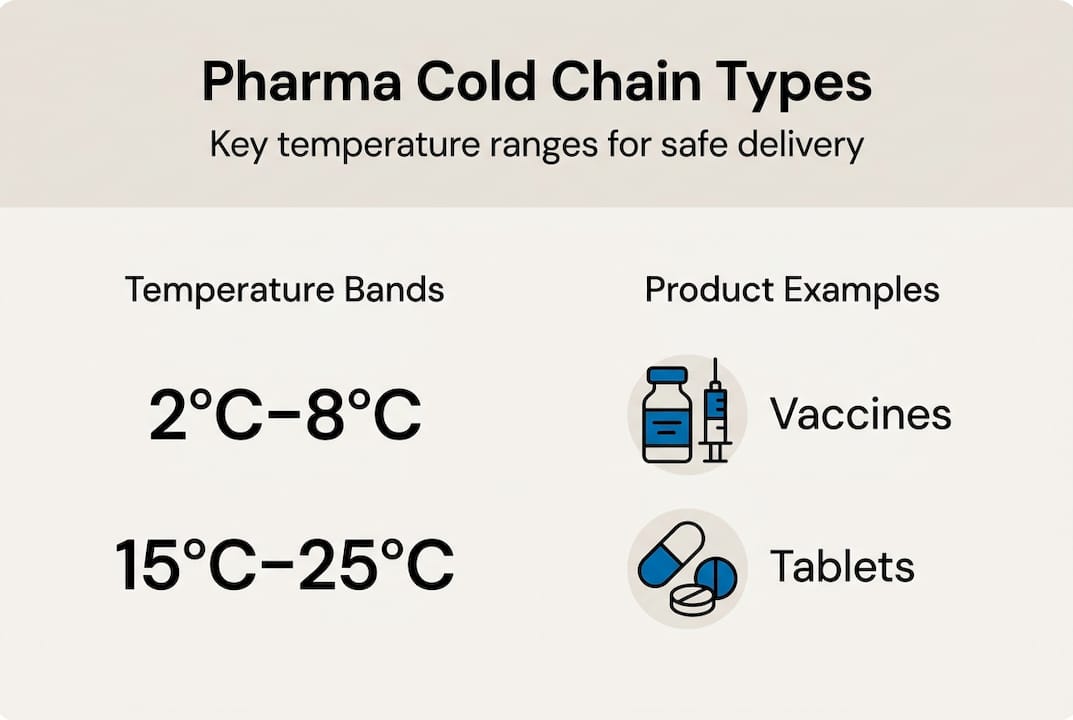
Standard temperature ranges for refrigerated products sit between 2°C and 8°C, with frozen products requiring -20°C and cryogenic storage dropping below -80°C. The precision demanded for mRNA vaccines — such as those developed for COVID-19 — brought this reality into sharp public focus, but the challenge applies across the entire biologics portfolio.
Here is how a temperature-sensitive pharmaceutical moves through its lifecycle:
- Manufacture — Product is formulated and quality tested under controlled conditions.
- Primary packaging and labeling — Sealed under validated environmental controls.
- Warehouse storage — Held in accredited, temperature-mapped cold rooms.
- Freight and customs — Transported in qualified containers with continuous monitoring.
- Regional distribution — Broken into smaller consignments and redistributed.
- Point-of-care storage — Held in validated refrigerators at clinics, hospitals, or pharmacies.
- Administration — Delivered to the patient within the approved storage window.
At any of these seven steps, a deviation can compromise the entire product history. Even a 30-minute exposure above 8°C for a refrigerated biologic can trigger a formal investigation and, in many cases, require product destruction.
“An unbroken cold chain is not a competitive advantage — it is a baseline requirement for patient safety and regulatory standing.”
Statistical data reinforces this. Temperature excursions are among the leading causes of pharmaceutical product recalls globally. For Southeast Asia, where ambient temperatures regularly exceed 30°C and humidity adds additional stress on packaging, maintaining these ranges is operationally demanding and non-negotiable.
Risks, edge cases, and compliance: Where the cold chain breaks
Understanding the standards is only part of the story. It is equally important to recognize where and how things go wrong in practice — especially in a region as diverse as Southeast Asia.
Edge cases in cold chain logistics include scenarios that standard SOPs often underestimate: power outages that cause refrigerators to exceed 8°C within 45 to 140 minutes, the freeze-thaw cycling of biologics during last-mile delivery, and deliberate shortcuts taken under time pressure during peak demand.
Key risk areas specific to Southeast Asia include:
- Power infrastructure gaps — Rural facilities in countries like Myanmar, Cambodia, and parts of Indonesia face frequent outages with no backup generation.
- Extreme ambient temperatures — Outdoor temperatures above 35°C accelerate excursions in uninsulated delivery vehicles.
- Geographic complexity — Island chains and mountainous terrain extend transit times dramatically, increasing exposure windows.
- Customs delays — Extended holding at ports without adequate cold storage adds unpredictable risk.
- Inadequate last-mile vehicles — Many final delivery vehicles lack validated temperature control, relying instead on passive cold boxes.
Last-mile challenges represent the most difficult segment to standardize, particularly for remote clinics and small hospitals that receive infrequent deliveries.
Research highlights a stark compliance reality: cold chain compliance under real operating conditions drops to as low as 88.8% in studies conducted across healthcare settings. That 11.2% non-compliance gap may sound small. But when applied across millions of doses distributed annually, it represents a significant volume of potentially compromised product.
Common pitfalls seen across pharma logistics challenges in the region include equipment that has not been serviced or recalibrated, staff who lack training on alarm response procedures, and documentation gaps that surface only during regulatory inspections.
Pro Tip: Redundant power systems (generator plus UPS backup) combined with continuous IoT-based temperature monitoring provide the strongest defense against excursions caused by infrastructure failures. Do not rely on manual checks alone.
Global standards and Southeast Asia’s regulatory landscape
To address real-world challenges, understanding the regulatory landscape and its demands is vital.
Good Distribution Practice (GDP) forms the backbone of pharmaceutical distribution compliance globally. In Southeast Asia, GDP requirements align with WHO and PIC/S (Pharmaceutical Inspection Co-operation Scheme) guidelines, which are progressively being harmonized across ASEAN markets to facilitate safer cross-border trade.
GDP compliance requires a set of verified practices including temperature mapping of storage facilities, real-time monitoring with validated equipment, redundant power systems, and qualified cold chain personnel. These are not optional elements — they are auditable requirements.
Key GDP compliance requirements include:
- Temperature mapping — Documented thermal surveys of all storage areas to identify hot and cold spots.
- Continuous monitoring — Automated data loggers with alert thresholds and calibration records.
- Redundant power — Generator backup with defined response time protocols.
- Qualified equipment — All refrigeration units and transport containers must be validated for their intended use.
- Staff training — Personnel must understand SOPs, deviation handling, and escalation procedures.
- Deviation management — Every excursion must be documented, investigated, and reported.
Regional harmonization efforts are advancing, with countries like Vietnam, Thailand, and Malaysia strengthening their regulatory frameworks to align with international benchmarks. However, gaps remain. Enforcement consistency across member states varies, and smaller distributors may lack the infrastructure to meet GDP requirements without significant investment.
“Harmonization creates consistency, but it cannot replace the physical infrastructure that standards demand. A documented GDP policy is meaningless without validated cold rooms, qualified transport, and trained personnel.”
For organizations entering or expanding within Southeast Asia, GDP-compliant cold chain services provided through accredited 3PL partners offer the most reliable path to compliance without requiring full in-house infrastructure buildout.
Why infrastructure, not just protocol, determines outcomes
There is a recurring debate in Southeast Asia’s pharmaceutical logistics sector: should companies invest in building in-house cold chain infrastructure, or outsource to specialist 3PL providers? The answer is less about cost and more about capability.
Protocols matter. GDP documentation matters. But the actual determinant of cold chain reliability is physical infrastructure. Backup power. Validated cold rooms. Calibrated sensors. Trained staff who respond correctly at 2 a.m. when an alarm triggers. These are not things that a well-written SOP can substitute for.
Many organizations focus intensely on ticking compliance checklists while underinvesting in the operational backbone those checklists are designed to protect. The checklist says “redundant power” — but does the generator actually start within 30 seconds? The SOP says “trained staff” — but has that training been tested under simulated failure conditions?
AI-driven cold chain monitoring is shifting the equation. Predictive analytics can flag equipment degradation before failure occurs, reducing excursion risk proactively rather than reactively. Smart 3PLs integrating these technologies offer a measurable reliability advantage over traditional approaches.
The uncomfortable truth for decision-makers is this: GDP compliance on paper and GDP performance in practice are two very different things. The organizations that close that gap are the ones investing in real infrastructure, not just documentation.
How to secure your cold chain: Strategic steps forward
Closing cold chain gaps requires more than policy updates. It demands the right operational partners, validated infrastructure, and continuous performance oversight.
Labgistics Asia brings over 20 years of specialized experience in pharmaceutical and life science logistics across Southeast Asia. From healthcare logistics support that streamlines regulatory compliance to smart warehousing solutions that integrate real-time inventory tracking, Labgistics provides the infrastructure that protocols alone cannot replace. For organizations committed to both compliance and sustainability, explore our eco-friendly cold chain options designed to reduce environmental impact without compromising product integrity. Every link in your cold chain deserves this level of precision.
Frequently asked questions
What is the cold chain and why is it needed for vaccines?
The cold chain is a temperature-controlled supply system essential for maintaining the safety and effectiveness of vaccines. Without it, vaccine integrity cannot be guaranteed from manufacture to patient administration.
How quickly does non-compliance impact medical product quality?
Pharmaceuticals can degrade irreversibly within minutes to hours of temperature exposure. Research shows that refrigerators can exceed safe temperature thresholds within 45 to 140 minutes of a power outage.
Which pharma products are most sensitive to temperature changes?
Vaccines, biologics, and monoclonal antibodies are among the most temperature-sensitive pharmaceutical products and require strict storage and transport controls.
What are good practices for cold chain compliance?
Essential practices include temperature mapping, real-time monitoring, redundant power systems, and the use of qualified cold chain equipment validated for pharmaceutical use.
Recommended
- Cold Chain Logistics for Pharma in Southeast Asia | Labgistics
- Green Cold Chain and Eco-Friendly Pharma Logistics | Labgistics
- AI in Cold Chain Logistics for Pharma | Labgistics
- Pharma Logistics Challenges and Smart Solutions | Labgistics
- Snelle verzending medicijnen: essentieel voor zorg
- Master laboratory supply chains for reliable reagents
