TL;DR:
- Southeast Asian lab supply chains face challenges like regulatory complexity, temperature-sensitive products, and infrastructure variability.
- Implementing real-time monitoring, integrated systems, and staff training is essential for effective optimization.
- Continuous management and technology integration are key to maintaining performance improvements over time.
Pharmaceutical and healthcare supply chain managers across Southeast Asia know the pressure well: a single temperature excursion, a missed regulatory filing, or a delayed shipment can wipe out thousands of dollars in product and set back critical timelines. Lab supply chains in this region carry the added weight of fragmented regulatory environments, extreme climate conditions, and rapidly growing demand. The cost of getting it wrong is not just financial. It touches patient safety, product integrity, and your organization’s reputation. This guide walks you through the core challenges, the tools you need, and a step-by-step process to systematically improve lab supply chain performance.
Table of Contents
- Understanding the challenges in lab supply chains
- Getting ready: Tools and requirements for lab supply chain optimization
- Step-by-step process: How to optimize your lab supply chain
- Troubleshooting and ensuring ongoing performance
- A practical perspective: Why most lab supply chain projects fail and what actually works
- Supercharge your lab supply chain with expert help
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Diagnose key challenges | Identifying unique pain points sets the stage for effective lab supply chain optimization. |
| Prepare with the right tools | Successful optimization requires the right mix of technology, training, and process alignment. |
| Follow proven steps | Systematic, step-by-step improvement yields lasting gains in efficiency and compliance. |
| Monitor and adapt | Continuous monitoring and troubleshooting keep your supply chain resilient and efficient over time. |
Understanding the challenges in lab supply chains
Now that we’ve framed how optimization can transform lab supply chain performance, let’s start by looking at the challenges unique to this sector.
Lab supply chains in Southeast Asia operate under conditions that would stress even the most mature logistics networks. You’re dealing with multi-country regulatory frameworks, high-value temperature-sensitive products, and infrastructure that varies widely from Singapore’s world-class facilities to more constrained environments in emerging markets. The result is a system prone to breakdowns at multiple points.
Common supply chain challenges in this sector include:
- Shipment delays caused by customs clearance bottlenecks, incomplete documentation, or carrier capacity issues
- Cold chain failures resulting from inadequate refrigeration during last-mile delivery or power interruptions at storage facilities
- Regulatory non-compliance stemming from inconsistent SOPs (standard operating procedures) across distribution partners
- Inventory inaccuracies driven by manual record-keeping and poor visibility into stock levels across multiple sites
- Staff skill gaps that lead to mishandling of sensitive samples or failure to follow validated protocols
The downstream impact of these failures is significant. Sample wastage, patient health risks, and cost overruns are the most visible consequences. Less visible but equally damaging are the regulatory penalties and the erosion of trust with clinical partners.
Consider a scenario that plays out more often than it should: a cold chain breach during a cross-border shipment from Thailand to Vietnam leads to the loss of a batch of diagnostic reagents. The replacement order takes two weeks. The clinical trial it was supporting is delayed. The cost of that single failure, when you add product loss, expedited freight, and regulatory reporting, can exceed the savings from an entire quarter of operational efficiency gains.
“Lab supply chains often stumble due to compliance lapses and temperature control failures.” This is not a warning to file away. It is a description of what happens when optimization is treated as optional rather than essential.
The compliance risks in pharma logistics are especially acute in Southeast Asia, where each country maintains its own import regulations, cold chain standards, and product registration requirements. Systematic optimization is the only reliable way to manage this complexity at scale.
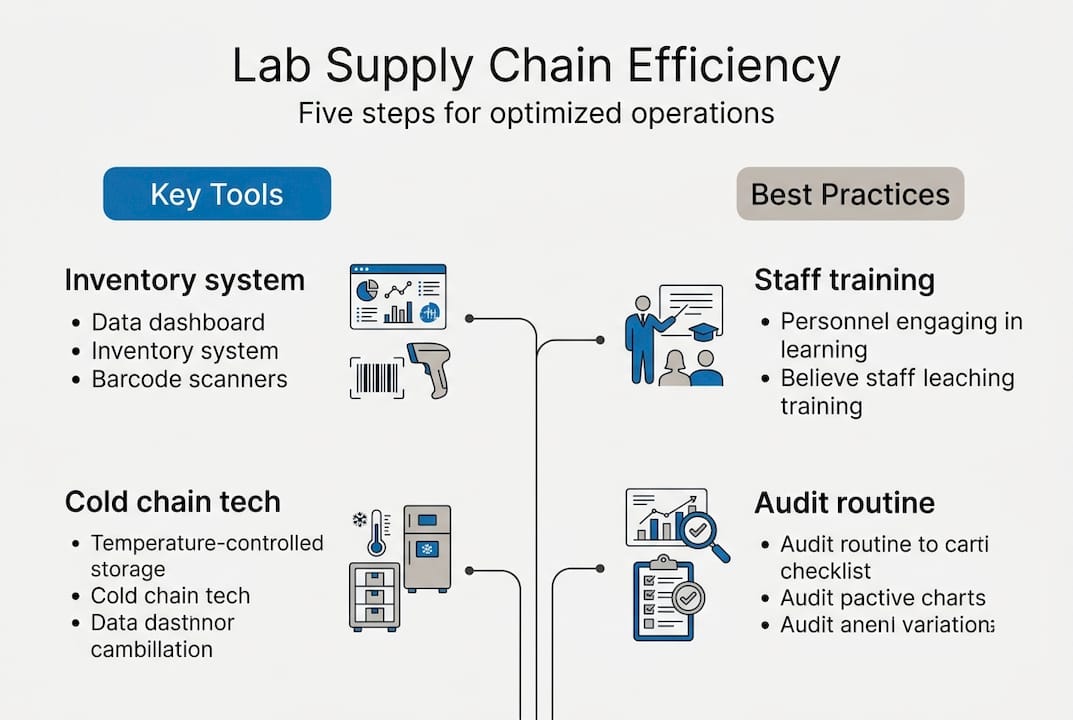
Getting ready: Tools and requirements for lab supply chain optimization
With the challenges identified, it’s time to gear up. Here are the prerequisites for a successful supply chain optimization project.
Before you can improve performance, you need the right infrastructure in place. Smart warehouse management and real-time inventory systems are not optional extras. They are the foundation on which every other improvement depends.

Here is a summary of the core tools and capabilities required:
| Capability | Purpose | Key consideration |
|---|---|---|
| Temperature monitoring systems | Detect and log excursions in real time | Must cover storage and transit |
| Inventory management software | Track stock levels, expiry dates, and movements | Integration with ERP systems preferred |
| Regulatory compliance protocols | Standardize documentation and SOPs across sites | Must align with each country’s requirements |
| Cold chain infrastructure | Maintain product integrity from origin to destination | Validation and calibration are mandatory |
| Trained logistics staff | Execute protocols accurately and consistently | Regular refresher training is essential |
Modern 3PL solutions and real-time inventory management are core enablers of this readiness. When selecting technology partners, prioritize vendors with proven experience in regulated healthcare environments. A vendor that serves general retail logistics will not understand the documentation requirements for temperature-sensitive pharmaceuticals.
Your baseline checklist before starting optimization:
- Conduct a gap analysis of current cold chain and inventory capabilities
- Map all regulatory requirements for each country in your distribution network
- Identify technology vendors with healthcare logistics credentials
- Secure executive sponsorship and cross-functional buy-in
- Establish baseline KPIs (key performance indicators) for delivery accuracy, temperature compliance, and inventory accuracy
Aligning your operations with sustainable practices in labs from the outset also reduces long-term costs and positions your organization well with regulators and partners who increasingly expect environmental accountability.
Pro Tip: Centralize all compliance documentation and staff training records in a single digital system before you begin. Teams that start with scattered paper records or siloed spreadsheets spend the first phase of any optimization project just catching up on basics, which delays results by weeks.
Step-by-step process: How to optimize your lab supply chain
Once your fundamentals are in place, it’s time to carry out the actual process of optimization step by step.
- Assess your current state. Audit every node in your supply chain: suppliers, warehouses, transport routes, and delivery points. Document where delays, errors, and compliance gaps occur most frequently.
- Map process bottlenecks. Use process mapping to visualize where handoffs break down. Focus on the intersections between teams, systems, and geographies.
- Implement the right tools. Deploy temperature monitoring, inventory management software, and compliance tracking systems. Prioritize integration so data flows automatically between platforms.
- Train your teams. Roll out structured training on new SOPs, technology platforms, and escalation procedures. Training is not a one-time event. Build it into your operational calendar.
- Monitor KPIs. Track delivery accuracy, cold chain compliance rates, inventory turnover, and regulatory audit outcomes. Review these metrics weekly during the initial phase.
- Drive continuous improvement. Use KPI data to identify remaining gaps. Run structured improvement cycles, adjusting processes and retraining staff as needed.
| Workflow element | Traditional approach | Optimized approach |
|---|---|---|
| Temperature tracking | Manual logs, periodic checks | Automated real-time alerts |
| Inventory management | Spreadsheets, reactive restocking | Software-driven, demand-based replenishment |
| Compliance documentation | Paper-based, site-specific | Centralized digital records, audit-ready |
| Staff training | Infrequent, informal | Scheduled, competency-verified |
| Performance review | Quarterly, reactive | Weekly, proactive |
Adopting integrated cold chain logistics drastically reduces loss rates and enhances regulatory compliance. This is especially relevant for cross-border shipments where temperature excursions are most likely to occur during handoffs between carriers.
For organizations ready to scale, AI in cold chain logistics introduces predictive capabilities that move you from reactive problem-solving to proactive risk management. AI-driven systems can flag potential excursions before they occur, based on route data, ambient conditions, and historical patterns.
Exploring pharma supply chain solutions tailored to the Southeast Asia region can also accelerate implementation by giving you access to validated frameworks rather than building from scratch.
Pro Tip: Digitize your compliance record-keeping from day one. Regulators in markets like Singapore, Malaysia, and Indonesia increasingly expect electronic audit trails. Organizations that maintain paper-based records face longer inspection cycles and higher risk of findings during regulatory audits.
Troubleshooting and ensuring ongoing performance
Even with an optimized supply chain, issues crop up. To maintain gains, you need robust monitoring and troubleshooting procedures.
Warning: A single compliance lapse, whether a missed temperature log, an expired certificate, or an undocumented deviation, can trigger a regulatory hold that stops your entire distribution operation. Prevention is always less costly than remediation.
The most common errors that surface after initial optimization include:
- Temperature excursions during last-mile delivery, often caused by driver behavior or equipment failure
- Inventory discrepancies between physical stock and system records, typically traced to manual receiving errors
- Documentation gaps in cross-border shipments where handoff records are incomplete
- Training drift, where staff revert to old habits after initial training without reinforcement
- Vendor non-compliance, where third-party carriers or suppliers fail to meet agreed standards
Spotting these issues early is the key to preventing escalation. Ongoing data review and temperature excursion alerts catch issues early and prevent costly product loss. Set up automated alerts for any deviation from defined thresholds, and assign clear ownership for investigation and resolution.
Your troubleshooting action list should include:
- Review temperature logs daily, not just when an alert fires
- Conduct unannounced spot checks on carrier cold chain equipment
- Reconcile inventory records against physical counts on a scheduled basis
- Run quarterly compliance audits across all distribution partners
- Maintain an escalation protocol so that any deviation reaches the right decision-maker within a defined timeframe
For organizations looking to reduce their environmental footprint while maintaining compliance, eco-friendly cold chains offer a framework for integrating sustainability into your monitoring and reporting systems without compromising product integrity.
AI-driven performance monitoring takes troubleshooting further by identifying patterns across large datasets that human reviewers would miss. When a specific route consistently shows minor excursions, AI flags it for investigation before a major failure occurs.
A practical perspective: Why most lab supply chain projects fail and what actually works
Now that you’ve seen the technical how-to, here’s a hands-on perspective on turning these plans into persistent success.
Most lab supply chain optimization projects do not fail because of bad technology choices. They fail because organizations treat optimization as a project with an end date rather than a continuous operational discipline. The technology gets deployed. The initial metrics improve. Then, six months later, performance quietly erodes because no one is driving the improvement cycle anymore.
The uncomfortable truth is that change management, not software, is the hardest part. Staff training that happens once and is never reinforced produces compliance drift. Audits that are scheduled rather than random lose their deterrent effect. Vendors that are onboarded without ongoing performance reviews gradually slip below standard.
What actually works is building optimization into your operational rhythm. Weekly KPI reviews, quarterly audits, and annual process revalidations are not bureaucratic overhead. They are the mechanism that keeps your gains from disappearing. Investing in sustainable warehousing strategies and regular process validation creates a system that improves itself over time rather than degrading after the initial push. Technology, people, and processes must evolve together. Focus your investment on change management and structured audits, not just tools.
Supercharge your lab supply chain with expert help
If you’re ready to see real results in supply chain efficiency and compliance, expert regional help is at hand.
Labgistics Asia brings over 20 years of specialized experience in pharmaceutical and healthcare logistics across Southeast Asia. From modern 3PL solutions and cold chain management to regulatory consulting and inventory optimization, the team delivers end-to-end support tailored to the complexity of this region. Whether you’re addressing smart solutions for pharma logistics or building a compliant distribution network from the ground up, Labgistics has the infrastructure, expertise, and regional relationships to accelerate your results. Reach out to schedule a supply chain assessment and discover where the greatest gains are within reach.
Frequently asked questions
What is lab supply chain optimization?
It’s the process of improving how materials, equipment, and samples flow through laboratory operations to reduce costs, delays, and errors. Optimization addresses operational inefficiencies and compliance challenges that are especially acute in regulated healthcare environments.
Why is cold chain management important for lab supply chains?
Cold chain management ensures temperature-sensitive lab samples and products remain viable, protecting patient safety and regulatory compliance. Proper cold chain logistics prevent sample loss and the compliance penalties that follow a documented excursion.
What are common errors in lab supply chain management?
Typical issues include inventory inaccuracies, compliance lapses, and shipment delays caused by manual record-keeping or inadequate staff training. Lab supply chains suffer from these compounding errors when there is no systematic monitoring in place.
How can technology improve lab supply chain efficiency?
Automation, real-time tracking, and AI-powered monitoring reduce errors, boost speed, and provide actionable insights for continuous improvement. AI and IoT solutions are transforming pharma supply chains by automating monitoring and reducing inefficiencies that manual systems cannot catch in time.
