Medical supply chains across Southeast Asia are under pressure that most risk registers simply do not capture. Geopolitical shocks, concentrated import routes, and a patchwork of national regulations have combined to expose costly vulnerabilities in the region’s pharmaceutical and medical device networks. For healthcare executives and supply chain managers, the stakes are direct: product shortages, compliance failures, and patient safety risks. This article identifies the most critical threats facing SEA supply chains today and presents evidence-backed mitigation strategies that go well beyond generic contingency planning.
Table of Contents
- Geopolitical and supply disruption risks
- Supplier concentration and import dependency
- Regulatory complexity and compliance barriers
- Risk assessment and resilience frameworks
- Building a responsive, sustainable supply chain
- What most supply chain leaders overlook about medical supply chain risks
- Mitigate supply chain risks with specialized healthcare logistics solutions
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Geopolitics amplify risks | Regional conflicts and chokepoints can disrupt essential medical supplies and drive up costs in Southeast Asia. |
| Import dependency is a weakness | Over-reliance on single countries for APIs and devices makes SEA supply chains vulnerable to shocks. |
| Regulatory complexity delays access | Inconsistent regulations across SEA countries hamper fast, efficient movement of medical goods. |
| Modern risk tools build resilience | Advanced frameworks and digital simulations help organizations identify and mitigate vulnerabilities proactively. |
| Sustainable practices safeguard supply | Blending resilience with sustainability and stakeholder collaboration strengthens long-term supply chain performance. |
Geopolitical and supply disruption risks
With the stakes of reliable supply chains established, let’s examine the greatest external threats: geopolitical and disruption-related risks.
Conflicts in West Asia and instability around critical maritime routes, including the Strait of Hormuz, have created measurable ripple effects across Southeast Asian healthcare supply chains. These are not abstract threats. Geopolitical disruptions cause real delays, cost surges, and acute shortages for essential medicines and medical devices reaching the region. When Indian pharmaceutical exporters face route disruptions, the downstream impact lands directly on hospital procurement teams in Bangkok, Kuala Lumpur, and Jakarta.
The cost consequences are significant. Disruptions to petrochemical supply lines have pushed prices for medical-grade plastics up by as much as 50%, affecting everything from IV bags to single-use device components. Specialty materials like helium, which is critical for MRI equipment and certain lab processes, face acute availability risks when key transit corridors are compromised. Anti-coagulants such as warfarin, partially sourced from Israeli manufacturers, have also appeared on shortage lists during recent conflict periods.
Key disruption scenarios that SEA supply chain leaders must plan for:
- Maritime route closures affecting pharmaceutical shipments from India and the Middle East
- Pandemic demand surges that simultaneously spike global consumption and reduce manufacturing output
- Climate events such as flooding in key production regions, disrupting raw material and finished goods availability
- Export restrictions imposed by source countries during domestic shortages
The country-level data reinforces the urgency. Thailand faces only 3 to 4 months of stock levels for certain critical medicines, while Malaysia relies on India for approximately 30% of its drug needs, creating concentrated exposure to any Indian supply disruption. For drug shortages in Malaysia, the risks extend beyond pharmaceuticals to medical devices and packaging materials.
“A supply chain built around a single import corridor is not a supply chain. It is a single point of failure with a long list of downstream consequences.”
Stay updated on emerging disruptions through pharma logistics news to maintain situational awareness before shortages escalate.
Supplier concentration and import dependency
Beyond geopolitics, the structural foundation of supply chains, the suppliers themselves, may be the greatest vulnerability.
China and India dominate the global supply of active pharmaceutical ingredients (APIs) and many categories of medical devices. The concentration is striking: 97.7% of certain antibiotics supplied to India originate from China, and India itself imports approximately 70% of its medical devices, with 20% of those coming from China alone. For Southeast Asian nations that rely on these two countries as primary sources, the risk is amplified significantly.
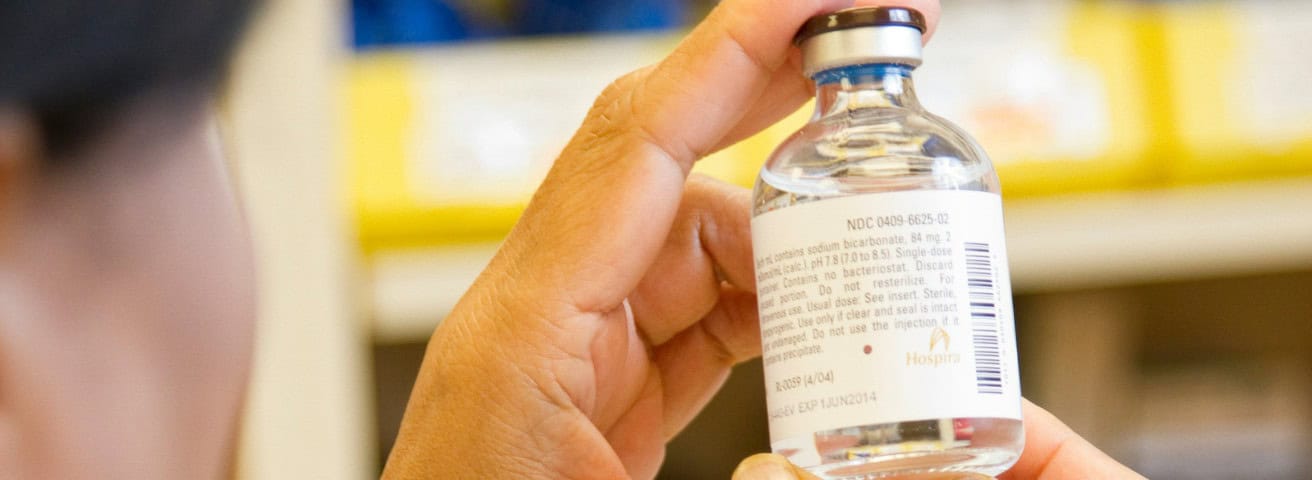
| Supply category | Primary source | SEA dependency level |
|---|---|---|
| Antibiotic APIs | China | Very high |
| Finished medical devices | India, China | High |
| Generic pharmaceuticals | India | High |
| Specialty raw materials | Multiple, concentrated | Moderate to high |
The practical consequences of this concentration include:
- Export bans during domestic shortages, as seen during COVID-19 when India restricted API exports
- Currency fluctuations that make import costs unpredictable for procurement teams working on fixed budgets
- Quality control gaps when rapid scaling by a single supplier compromises batch consistency
- Regulatory retaliation risks when bilateral trade tensions affect healthcare goods
For SEA healthcare organizations, pharma logistics challenges rooted in supplier concentration require structural solutions, not just reactive purchasing. The pharma supply chain vulnerabilities exposed by the West Asia conflict have accelerated conversations about regional manufacturing and alternative sourcing.
Pro Tip: Implement a multi-sourcing policy that qualifies at least two geographically distinct suppliers for every critical API and device category. Pair this with a regional supplier development program that supports ASEAN-based manufacturers in meeting international quality standards.
Regulatory complexity and compliance barriers
Complex supplier networks are further challenged by a patchwork of regulatory regimes that slow and complicate the movement of critical products.
The ASEAN Medical Device Directive (AMDD) represents meaningful progress toward regional harmonization, but it reduces rather than eliminates regulatory complexity. Each member state maintains its own National Regulatory Authority (NRA), classification system, and dossier requirements. The ASEAN regulatory framework uses a four-tier classification system (Class A through D), but the practical application of these classifications varies by country, creating compliance uncertainty for manufacturers and distributors.
| Country | Regulatory body | Key requirement | Typical approval timeline |
|---|---|---|---|
| Singapore | HSA | ASEAN Common Submission Dossier | 30 to 270 days |
| Malaysia | MDA | Local authorized representative | 90 to 365 days |
| Thailand | FDA Thailand | Thai-language labeling, local rep | 180 to 540 days |
For medical device companies, local representative requirements mean that market access depends on establishing in-country partnerships before a single unit ships. ISO 13485 certification is often mandatory, and country-specific post-market surveillance obligations add ongoing compliance costs.
Steps to reduce regulatory risk across SEA markets:
- Map regulatory requirements for each target market before product launch, not after
- Engage a regional regulatory affairs specialist who maintains active relationships with local NRAs
- Standardize technical documentation to align with ASEAN Common Submission Dossier templates
- Build post-market surveillance SOPs that can be adapted for each country’s reporting requirements
- Monitor Malaysian reporting on regulatory risk and equivalent national updates regularly
Understanding regulatory market entry through Singapore as a regional hub can significantly reduce time-to-market. Pairing this with smart 3PL inventory management ensures compliant storage from day one.
Risk assessment and resilience frameworks
To shift from reactive problem-solving to strategic risk leadership, a new toolkit for assessment and resilience is essential.
Many SEA healthcare organizations still rely on static risk registers and annual supplier audits. These tools are necessary but insufficient. Modern risk management requires dynamic, quantitative approaches that can model cascading failures and test assumptions before a crisis occurs.
Probabilistic Risk Assessment (PRA) and its pharmaceutical-specific variant, SUPRA (Supply chain Probabilistic Risk and Uncertainty Assessment), allow organizations to assign likelihood and impact scores to specific disruption scenarios. Agent-based simulation goes further, modeling how individual supplier decisions and logistics events interact across an entire network. These tools give supply chain leaders a quantified view of where their greatest exposures actually sit.
Key components of a robust resilience framework:
- Multi-criteria decision models (MCDM) for supplier evaluation, incorporating financial stability, geographic risk, quality history, and sustainability performance
- Supplier audits structured around compliance KPIs and environmental, social, and governance (ESG) criteria
- Buffer stock policies calibrated separately for generic drugs, specialty pharmaceuticals, and medical devices based on criticality and lead time
- Redundant sourcing agreements that are pre-qualified and ready to activate, not just listed on a spreadsheet
For supply chain resilience practices, integrating internal operational data with external supply intelligence creates a more complete risk picture. Calibration and validation best practices also play a direct role in resilience by ensuring that storage and handling conditions remain verified under stress conditions. Supply resilience frameworks from academic and industry sources increasingly emphasize stakeholder engagement as a core resilience driver.
Pro Tip: Run at least one scenario stress-test per quarter using a real disruption event from the past 12 months as your input. This keeps risk models current and builds organizational muscle memory for crisis response.
Building a responsive, sustainable supply chain
The most resilient organizations embed these tools in daily operations, building flexibility and sustainability into their culture and systems.
Scenario planning is the bridge between risk assessment and operational readiness. When supply chain teams regularly rehearse disruption scenarios, including port closures, supplier insolvencies, and regulatory holds, they make faster and better decisions when real events occur. Digital platforms that provide real-time visibility into inventory levels, shipment status, and supplier performance are now a baseline requirement, not a differentiator.
Sustainability and resilience are increasingly linked. Sustainability-resilience frameworks supported by WHO data show that supply chains designed with environmental and social responsibility in mind also perform better during disruptions. Diversified supplier bases, lower carbon logistics routes, and reduced single-use packaging all contribute to both goals simultaneously.
An action checklist for SEA supply chain leaders:
- Conduct quarterly risk reviews that incorporate geopolitical, regulatory, and supplier-level updates
- Maintain buffer inventory for all Category A and B critical items, with defined reorder triggers
- Diversify logistics providers to avoid single-carrier dependency for critical shipments
- Engage procurement, quality, regulatory, and logistics teams in a shared risk governance structure
- Benchmark against stakeholder-inclusive frameworks that reflect current WHO SEA regional priorities
For sustainable medical supply chains, the goal is not just continuity during crisis but consistent, high-quality performance across normal operations. Organizations that integrate resilience into procurement policy, supplier contracts, and logistics SOPs outperform those that treat it as a separate risk management function.
What most supply chain leaders overlook about medical supply chain risks
Despite the wealth of tools available, many organizations still repeat the most avoidable mistakes. The most common error is focusing risk management attention almost entirely on immediate, tier-one suppliers while ignoring the geopolitical, regulatory, and environmental forces that operate several layers upstream. A disruption to a Chinese API manufacturer affects your Indian finished-goods supplier before it affects you, but by the time it reaches your inventory, the response window has closed.
Static risk maps are another persistent weakness. A risk register that is updated annually cannot keep pace with the speed at which geopolitical and regulatory conditions shift in Southeast Asia. Stress-testing with real, recent scenarios is far more valuable than theoretical heat maps. Organizations that run live disruption exercises consistently respond faster and with less financial damage.
Regional collaboration is underutilized. Sharing supply intelligence with peer organizations, engaging with ASEAN-level regulatory bodies, and building relationships with alternative logistics partners before you need them are all practices that separate genuinely resilient supply chains from those that simply look resilient on paper. Deeper sustainability insights consistently show that cross-functional, cross-border coordination is the single greatest predictor of supply chain performance under pressure. Act before the crisis, not after.
Mitigate supply chain risks with specialized healthcare logistics solutions
Turning these strategies into reality is easier with experienced, technology-led logistics partners.
Labgistics Asia brings over 20 years of specialized healthcare logistics experience across Southeast Asia, with infrastructure and expertise designed specifically for the risks outlined in this article. From smart medical warehousing with real-time inventory visibility to cold chain management for temperature-sensitive pharmaceuticals, every service is built around regulatory compliance and supply continuity.
For organizations navigating logistics challenges solutions across multiple SEA markets, Labgistics provides end-to-end 3PL cold chain support that integrates warehousing, distribution, and regulatory support into a single, accountable partnership. Contact Labgistics Asia to assess how your current supply chain strategy aligns with the region’s evolving risk landscape.
Frequently asked questions
What are the biggest medical supply chain risks in Southeast Asia?
The top risks are geopolitical disruptions, over-reliance on a few countries for imports, regulatory fragmentation, and lack of robust risk assessment frameworks. Geopolitical disruptions and import dependency are particularly severe for SEA medical supply chains given the region’s concentrated sourcing patterns.
How can healthcare executives mitigate supply chain risks?
Key mitigation steps include multi-sourcing, buffer stock, scenario planning, and working with logistics partners who understand regional regulations. Multi-sourcing and scenario planning are recognized as foundational best practices for building regional supply chain resilience.
Why is regulatory compliance difficult across Southeast Asia?
Each country enforces unique requirements, classifications, and dossier formats despite ASEAN efforts, which raises costs and causes market delays for medical devices and pharmaceuticals. The ASEAN regulatory framework reduces but does not eliminate this complexity.
Which products are most at risk of supply disruption?
Products highly dependent on a single origin, like vital antibiotics, anti-coagulants, and specialized materials such as plastics and helium, are most vulnerable. Edge case products like warfarin and helium exemplify how supply shocks can affect even narrowly sourced inputs.
What role does technology play in supply chain risk management?
Advanced analytics, digital platforms, and simulation models enable early detection, scenario planning, and more resilient inventory management. Agent-based simulation and PRA improve risk readiness by modeling cascading failures before they occur in real operations.
