Regulatory Services
Labgistics provides end-to-end regulatory affairs services that enable our clients to register medical devices and pharmaceutical products across different countries.
With a specialised team of regulatory affairs experts and over 1,000 completed registrations across Asian markets, we provide customised and accelerated solutions to help your products achieve market access while meeting all necessary regulatory requirements for registration, import, and supply.
Our regulatory team provides support for your company throughout the product lifecycle, ensuring compliance with regulations through vital and strategic advice from product development to commercialisation.

Our Services
Registration
Launch
Post Launch
We offer a comprehensive suite of end-to-end regulatory services, covering medical devices, therapeutic products, active ingredients, radioactive materials, poisons, and controlled drugs. Each service is tailored to meet our clients’ specific requirements.
Our experienced team understands the complexities of the regulatory landscape and works closely with clients to ensure compliance and uphold the highest safety standards for every product. Whether you require support for radioactive transportation or access to our distribution centre, you can explore our full range of regulatory services below.
What Kind of Service Are You Looking For?
Regulatory Intelligence
Pre-Submission Capabilities
Consultation with Health Authorities
Product Registrations and Submissions
Pharmacovigilance Expertise
Labelling and Packaging Compliance
Post Approval Services
Country-Specific Regulatory Information
Risk Classification
In Singapore, there are four risk classes of medical devices.
General Medical Devices
| Class | Risk Level |
|---|---|
| A | Low Risk |
| B | Low-moderate Risk |
| C | Moderate-high Risk |
| D | High Risk |
In-Vitro Diagnostic Medical Devices
| Class | Risk Level |
|---|---|
| A | Low Individual Risk and Low Public Health Risk |
| B | Moderate Individual Risk and/or Low Public Health Risk |
| C | High Individual Risk and/or Moderate Public Health Risk |
| D | High Individual Risk and High Public Health Risk |
Software as Medical Devices (SaMD)
Non-IVD SaMD
Risk Classification Table
| State of healthcare situation or condition | Significance of information provided by SaMD to healthcare decision | ||
|---|---|---|---|
| Treat or diagnose | Drive clinical / patient management | Inform clinical / patient management | |
| Critical | C | C | B |
| Serious | C | B | A |
| Non-serious | B | A* | A |
Class A medical devices:
Exempted from registration but requires listing/product notification on HSA Class A database prior to import and supply
Product registration or Special Access Route (SAR) is required prior to import and supply.
Registration
Type of Applications
- NDA-1: For the first strength of a product containing a new chemical/biological entity
- NDA-2: For the first strength of a product
- NDA-3: For subsequent strength(s) of a product that has been registered or has been submitted as an NDA-1 or NDA-2
Generic drug application
- GDA-1: For the first strength of a generic chemical product.
- GDA-2: For subsequent strength(s) of the generic chemical product that has been registered or submitted as GDA-1.
Risk Classification
Medical devices are generally categorized into two main groups based on their intended purpose and use: General Medical Devices (used in direct contact with the body for treatment or diagnosis) and In Vitro Diagnostic (IVD) Medical Devices (used outside the body on samples derived from it).
Both categories are classified by risk—usually Class A/I (low) to D/III or IV (high)—to determine regulatory requirements.
General Medical Devices
Table 1: Classification system for General Medical Devices
| Class | Risk Level |
|---|---|
| A | Low Risk |
| B | Low-moderate Risk |
| C | Moderate-high Risk |
| D | High Risk |
Table 1: Classification system for IVD Medical Devices
| Class | Risk Level |
|---|---|
| A | Low Individual Risk and Low Public Health Risk |
| B | Moderate Individual Risk and/or Low Public Health Risk |
| C | High Individual Risk and/or Moderate Public Health Risk |
| D | High Individual Risk and High Public Health Risk |
General Principles of Grouping
General medical devices can be grouped into one of the following four categories and can be submitted in one application for medical device registration:
a) SINGLE
b) SYSTEM
c) FAMILY
d) SET
In-Vitro Diagnostic Medical Devices
In-Vitro Diagnostic Medical devices that can be grouped into one of the following five categories can be submitted in one application for medical device registration and listing in the Malaysia Medical Device Register (MMDR):
a) SINGLE
b) SYSTEM
c) FAMILY
d) IVD TEST KIT
e) IVD CLUSTER
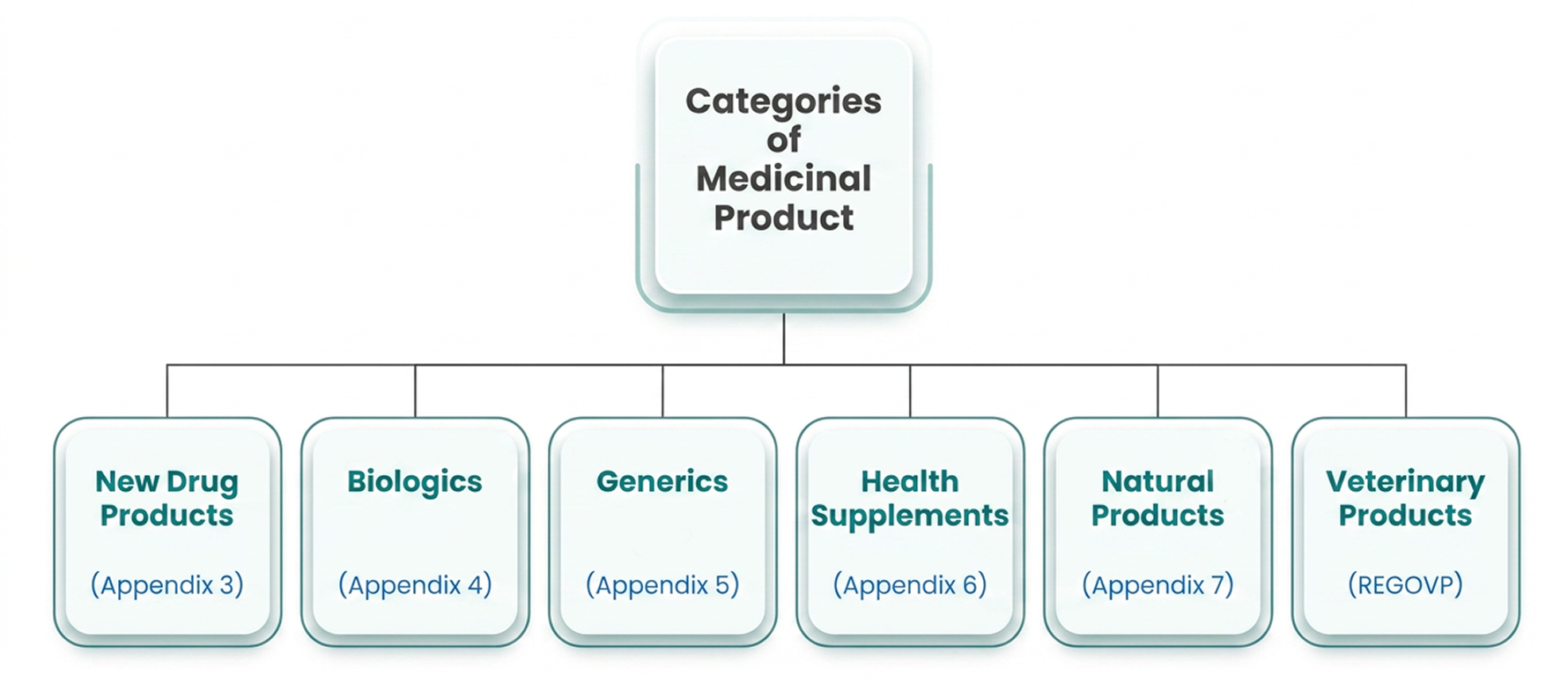
Registration
Type of Applications
Risk Classification
Groups of medical devices are divided into 2 groups for IVDs and Non IVDs.
In-Vitro Diagnostic Medical Devices (IVD)
Medical devices including instruments, machines, reagents, software, or other products intended by the manufacturer for medical or diagnostic use, including providing testing result from human specimens, used alone or in combination, and whose primary action is not achieved by pharmacological, immunological, or metabolic means.
Table 1: Classification system for General Medical Devices
| Class | Risk Level |
|---|---|
| A | Low individual and public health risks |
| B | Moderate individual risk or a low public health risk |
| C | High individual risk or a moderate public health risk |
| D | High individual and public health risks |
Non-IVD (General medical device & Active medical device)
General Medical Device
Medical device which is not In Vitro Diagnostic (IVDs) products.Active Medical Device
- Active Medical Device means a medical device that depends on a source of electrical energy or other external energy, other than that generated by the human body or gravity, to operate.
- Specific rule for Software as medical device (SaMD) classification & AI medical device/ Machine Learning (ML)
| Class | Risk Level |
|---|---|
| A | Low |
| B | Low-Moderate |
| C | Moderate-High |
| D | High |
Prohibited Medical Device
Prohibited medical devices which can’t be manufactured, imported, or sold.- Alloy and Mercury Products used to blend Dental Amalgam for dental filling in tablet/ powder forms (only Sachet form is allowed)
- Ear candle
- Surgical gloves with powder
- Macrotextured rough skin type implanted breast silicone (Average surface roughness > 50 𝜇𝑚 following non-active surgical implants-Mammary Implants-Particular requirements-ISO 14607:12018)
Registration
Type of Applications
New drug application
New medicinal product refers to drugs with their own research and development
Generic drug application
Generic medicinal product (Well-established medicine) – New generic medicinal product
Risk Classification
Medical Devices
The classification of a medical device or multiple medical devices must rely on the rules for classification using A, B, C, D levels of risks.
Table 1: Classification system for Medical Devices
| Class | Risk Level |
|---|---|
| A | Low Risk |
| B | Low-moderate Risk |
| C | Moderate-high Risk |
| D | High Risk |
Registration
Type of Applications
- New chemical drug, Vaccine, Biologicals
- New chemical drug but not brand-name drug
- Chemical drugs which have the same drug substances, strength, concentration, administration route and dosage form as those of drugs granted marketing authorization by one of the drug regulatory authorities SRA countries but do not have original brand-name drugs granted marketing authorization in Vietnam or whose strength, concentration, administration route or dosage form is different from that of original brand-name drugs granted marketing authorization in Vietnam
- Generic
Country-Specific Regulatory Information
Risk Classification
In Singapore, there are four risk classes of medical devices. The risk classification of a medical device is dependent on:
General Medical Devices
Depends on a number of factors eg. Duration of medical device contact with body, degree of invasiveness, whether the medical device delivers medicinal products or energy to the patient, whether they are intended to have a biological effect on the patient and local versus systemic effects.
| Class | Risk Level | Example |
|---|---|---|
| A | Low Risk | Wheelchairs, Tongue depressors |
| B | Low-moderate Risk | Hypodermic needles, Suction equipment |
| C | Moderate-high Risk | Ventilators, Bone fixation plates |
| D | High Risk | Heart valves, Implantable defibrillators |
Table 1: Classification system for General Medical Devices
In-Vitro Diagnostic Medical Devices
Depends on intended purpose, technical expertise of intended user (lay person or healthcare professional), importance of information to the diagnosis (sole determinant or one of several), impact of result to the individual and/or to public health.
| Class | Risk Level | Example |
|---|---|---|
| A | Low Individual Risk and Low Public Health Risk | Specimen receptacle |
| B | Moderate Individual Risk and/or Low Public Health Risk | Vitamin B12, Pregnancy self testing, Anti-Nuclear Antibody, Urine test strips |
| C | High Individual Risk and/or Moderate Public Health Risk | Blood glucose self testing, HLA typing, PSA screening, Rubella IgM |
| D | High Individual Risk and High Public Health Risk | HIV blood donor screening, HIV diagnostic kit |
Table 2: Classification system for IVD Medical Devices
Software as Medical Devices (SaMD)
Depends on state of patient’s healthcare situation or condition, significance of information provided by SaMD to healthcare decision.
Non-IVD SaMD Risk Classification Table
| State of healthcare situation or condition | Significance of information provided by SaMD to healthcare decision | ||
|---|---|---|---|
| Treat or diagnose | Drive clinical / patient management | Inform clinical / patient management | |
| Critical | C | C | B |
| Serious | C | B | A |
| Non-serious | B | A* | A |
*SaMD will be classified as Class B if they are intended to analyse, measure or monitor a vital physiological process or anatomical structure images
| Forms of Software | Examples |
|---|---|
| Software embedded in medical devices |
|
| Standalone software |
|
| Standalone mobile applications |
|
| Web-based software |
|
HSA Medical device risk classification tool
Class A medical devices:
Exempted from registration but requires listing/product notification on HSA Class A database prior to import and supply
Product registration or Special Access Route (SAR) is required prior to import and supply.
Registration
Determine Risk Classification
Determine if the medical device is a Class A, B, C or D medical device based on HSA’s criteria
Determine Type of Registration Evaluation Route
- No prior approval from HSA’s reference agencies
- At least one approval from HSA’s reference agencies
- At least one approval from HSA’s reference agencies and
- 3 years marketing history without any safety issue, withdrawal, or rejection (IBR Condition 1)
- At least two approvals from HSA’s reference agencies and
- No safety issues, withdrawal, or rejection (IBR Condition 2)
- At least one approval from HSA’s reference agencies and
- 3 years marketing history without any safety issue, withdrawal, or rejection (ECR-1)
- At least two approvals from HSA’s reference agencies and
- No withdrawal or rejection (ECR-2/EDR)
- At least one approval from HSA’s reference agencies and
- No safety issues, withdrawal, or rejection
| No. | RA Authorities | Reference Countries |
|---|---|---|
| 1 | United States Food and Drug Administration (US FDA)
| USA |
| 2 | Australia Therapeutic Goods Administration (TGA) Device Registration Licence | Australia |
| 3 | Health Canada (HC) Device Registration Licence | Canada |
| 4 | Japan Ministry of Health, Labour and Welfare (MHLW)
| Japan |
| 5 | European Union Notified Bodies (EU NB)EC Certificates issued according to: Class B:
Class C & D:
| European Union |
Class B, C and D medical devices that are classified and approved/cleared as Class I or class II exempt in the above reference agencies do not qualify for abridged, expedited or immediate routes based on these approvals.
Collect Documents for Registration
| No. | DOCUMENTS (softcopy) | HSA Template | Remarks |
|---|---|---|---|
| 1 | Letter of authorization (Annex 1) | ✔ | |
| 2 | Annex 2 List of Configurations | ✔ | |
| 3 | Proof of reference agency’s approval | Not required for full route | |
| 4 | Proof of marketing history (Annex 2) | ✔ | Required for IBR (Condition 1) & ECR-1 |
| 5 | Declaration of no safety issues globally (Annex 3) | ✔ | Required for IBR, ECR-1 & ICR |
| 6 | Executive Summary | ||
| 7 | Essential principles checklist and Declaration of conformity | ✔ | Not required for IBR & ICR |
| 8 | Device description | ||
| 9 | Design verification and validation documents | Not required for IBR route | |
| 10 | Clinical evidence | ||
| 11 | Proposed device labelling | ||
| 12 | Risk analysis | Not required for IBR & ICR | |
| 13 | Manufacturer Information (site’s name and address) | ||
| 14 | Proof of QMS (eg. ISO13485/MDSAP certificate etc) | ||
| 15 | Manufacturing Process – flow chart | Not required for IBR, ICR & Class B Abridged | |
| 16 | Justification for an unmet clinical need | Only required for Priority review scheme 1 |
For SaMD, there are additional specific requirements for the following sections of the registration dossier:
- Essential Principles for safety and performance of medical devices
- Labelling requirements
- Software versioning and traceability
- Software verification and validation
- Clinical Evidence
- Risk Management
- Supporting documents for Cybersecurity
Additional requirements for Artificial Intelligence Medical Devices (AI-MD):
The following additional regulatory considerations will be applicable to medical services incorporating Artificial Intelligence (AI) technology from a medical device regulatory standpoint.
The following additional information are required to be submitted for pre-market registration of AI-MDs, in addition to the list of documents stated above:
- AI-MD Dataset
- AI-MD Model
- AI-MD Performance and Clinical Evaluation
- AI-MD Deployment
Submit registration to HSA and address input requests until approval
Submit complete registration dossier to HSA and work closely with product owner to address potential input requests from HSA until product registration approval is obtained.
Registration
Determine Risk Classification
New drug application
- NDA-1: For the first strength of a product containing a new chemical/biological entity
- NDA-2: For the first strength of a product
- containing a new combination of registered chemical or biological entities
- containing registered chemical or biological entity(ies) in a new dosage form, new presentation, or new formulation
- containing registered chemical or biological entity(ies) for use by a new route of administration or
- containing registered chemical or biological entity(ies) for new indication(s), dosage recommendation(s) and/or patient population(s).
- NDA-3: For subsequent strength(s) of a product that has been registered or has been submitted as an NDA-1 or NDA-2
Generic drug application
- GDA-1: For the first strength of a generic chemical product.
- GDA-2: For subsequent strength(s) of the generic chemical product that has been registered or submitted as GDA-1.
Determine Type of Registration Evaluation Route
- Full (NDA): No prior approval from any regulatory agencies
- Abridged (NDA/GDA): Approval from at least one drug regulatory agency
- Verification (NDA/GDA): Approval from at least two of HSA’s reference agencies
- Verification-CECA (GDA): Applies to any generic product manufactured in India which has approval from HSA’s reference agencies
List of HSA’s reference regulatory agencies:
| No. | RA Authorities | Reference |
|---|---|---|
| 1 | United States Food and Drug Administration (US FDA) | USA |
| 2 | Australia Therapeutic Goods Administration (TGA) | Australia |
| 3 | Health Canada (HC) | Canada |
| 4 | UK Medicines and Healthcare Products Regulatory Agency (UK MHRA) via national procedure or as Reference Member State via Mutual Recognition Procedure or Decentralised Procedure on or prior to 31 January 2020 | United Kingdom |
| 5 | European Medicines Agency (EMA) via the Centralised Procedure | European Union |
| 6 | Swissmedic | Switzerland |
Collect Documents for Registration
| No. | Documents | Location in | |
|---|---|---|---|
| ICH CTD | ACTD | ||
| 1 | Administrative Documents and Product Information | Module 1 | Part I |
| 2 | Common Technical Document Overview and Summaries | Module 2 | Incorporated in Parts II, III and IV |
| 3 | Quality documents | Module 3 | Part II |
| 4 | Non-clinical documents | Module 4 | Part III |
| 5 | Clinical documents | Module 5 | Part IV |
